Graphene Oxide Synthesis by using Graphite, Sulphuric Acid, Potassium Permanganate and Hydrogen Peroxide via Modified Hummer’s Method
Cite This in Your Publication
Graphene Oxide Synthesis by using Graphite, Sulphuric Acid, Potassium Permanganate and Hydrogen Peroxide via Modified Hummer’s Method - InstaNANO. https://instanano.com/all/nanomaterial-synthesis/carbon/graphene-oxide-1/ (accessed May 3rd, 2024).
Graphene Oxide Synthesis by using Graphite, Sulphuric Acid, Potassium Permanganate and Hydrogen Peroxide via Modified Hummer’s Method - InstaNANO. https://instanano.com/all/nanomaterial-synthesis/carbon/graphene-oxide-1/ (accessed May 3rd, 2024).
Graphene Oxide Synthesis by using Graphite as precursor, Sulphuric Acid and Potassium Permanganate as Oxidising Agent and Hydrogen Peroxide via Modified Hummer’s Method
-
CHECK LISTGraphite, Sulphuric Acid, Potassium Permanganate (KMnO4), Hydrogen Peroxide (H2O2), Deionized Water, RB Flask, Magnetic Stirrer
-
STEP 1.Add 1g Graphite into 25ml Sulphuric acid and setup ice-bath.
-
STEP 2.Add 3g Potassium Permanganate very slowly by keeping the temperature less than 20ºC and stir for more 3 hours.
NOTE: You may remove the ice bath after 20 minutes and again setup for step-3
-
STEP 3.Add 50ml water drop-wise by keeping the temperature less than 50ºC.
This is very dangerous step take proper precautions
-
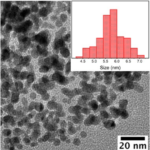
STEP 4.After some time the color of colloidal changes to dark brown indicates the formation of Graphene Oxide
-
STEP 5.Now, add 100ml water instantly for complete oxidation.
-
STEP 6.Add 5ml Hydrogen Peroxide for the completion of the reaction.
-
STEP 7.Washing Process: Add 100ml Hydrochloric Acid in 900ml deionized water and wash Graphene Oxide (produced after step-6) with it several times for 15-20 minutes each at 5000rpm.
-
STEP 8.Wash again with deionized water 5-6 times for 30 minutes each at 6000rpm.
-
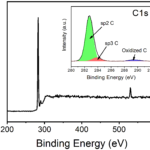
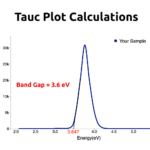
RESULTSGraphene Oxide (GO) colour would be dark brown; UV-VIs spectroscopy absorbance peak would be obtained around 250nm; Raman spectroscopy D band and G band would be obtained at around 1300cm-1 and 1550cm-1 respectively.
-
Factors Affecting SynthesisConcentration of Graphite: The amount of Graphite used for the synthesis of Graphene Oxide (GO) is very important factor; because oxidation penetration on Graphite depends on the concentration of the Graphite in the Sulphuric Acid. Higher the concentration of Graphite, lesser the penetration of oxidation.
Concentration of Sulphuric Acid: Concentration of Sulphuric Acid is most important in Hummers method. Higher the concentration of Sulphuric Acid, higher the chances of complete exfoliation of Graphite into Graphene Oxide; But also very high chances of unwanted accident.
Rate of Adding Water: Rate at which water is added for the oxidation matters a lot. InstaNANO suggest you to add water drop wise very slowly to avoid unwanted accidents and to get good quality Graphene Oxide.
Effect of KMnO4: In this method, KMnO4 acts as Oxidizer, Sulphuric Acid functionalize the Graphite surface to add functional groups in the presence of reagent like KMnO4. KMnO4 should be added slowly because it increases the temperature of the reaction and may lead to incomplete oxidation at the end, if poured instantly and not stirred properly.
Role of Hydrogen Peroxide: Main role of Hydrogen Peroxide is to remove the excessive KMnO4 or in other words to stop the reaction.
Concentration of HCL: Hydrochloric Acid (HCL) is used in the washing process of Graphene Oxide (GO) because HCL can remove/dissolve any metal impurity during the synthesis of GO from Hummers method. By using concentrated HCL partial reduction of GO may be possible.
Centrifuge Time and Speed: Centrifuge time and speed (rpm) decide the amount of GO settle down in the centrifuge tube. At higher speed and more time washing, even smaller size GO can settle down in the centrifuge tube.
Number of Washing: How many times you wash is very important in this washing process. More times you wash the GO, leads to good quality GO with less impurities. -
NOTE: All the experiments should be done under the guidance of lab Incharge; and proper lab safety instructions.






















During synthesis of graphene oxide my sample turns out green after adding DI water and Hydrogen Peroxide instead yellow..How can I remove this
i refluxed the sample after ice bath at 80 degree celsius, after that when i added hydrogen peroxide , it completely turned black not yellow or transparent. What should i do?
Did you manage to solve that problem?
Please check all your chemicals that they are not expired and also try with some other graphite powder (graphite powder source is highly relative to the quality of GO). You should also add water slowly in step-3 as this can also change everything.